 The review also identified nearly 500 deaths linked to the devices, along with nearly 78,000 injuries and 30,000 malfunctions. An FDA review of adverse events involving stimulators found that nearly a third were reports of poor pain relief. A recent study found that many patients with stimulators did not reduce their use of opioids, epidurals, corticosteroid injections or radiofrequency ablation and about one in five had complications so severe the devices had to be removed or revised.Ī 2018 study found that SCSs have some of the worst safety records of medical devices tracked by the FDA. and their use is growing – in part because of the belief they’ll reduce the need for opioids and other pain therapies. In no event shall Boston Scientific be liable for any incidental, special, or consequential loss, damage, or expense, direct or indirect, related to the purchase. :max_bytes(150000):strip_icc()/spectra-5a22d02beb4d52001a7722a1.jpg)
The devices are surgically implanted near the spine or brain, and emit low-level electrical impulses to block pain signals.Ībout 50,000 spinal cord stimulators (SCSs) are implanted annually in the U.S. Boston Scientific disclaims any and all warranties, expressed or implied, including any implied warranty of merchantability or fitness for a particular purpose or application. Implantable neurostimulators are an invasive treatment of last resort for people with chronic back, leg or head pain. Proclaim XR 7 IPG Model 3662 (Previously known as the ‘Proclaim 7 Elite IPG Model 3662’) Proclaim XR 5 IPG Model 3660 (previously known as the ‘Proclaim 5 Elite IPG Model 3660’) The following devices are included in the recall:  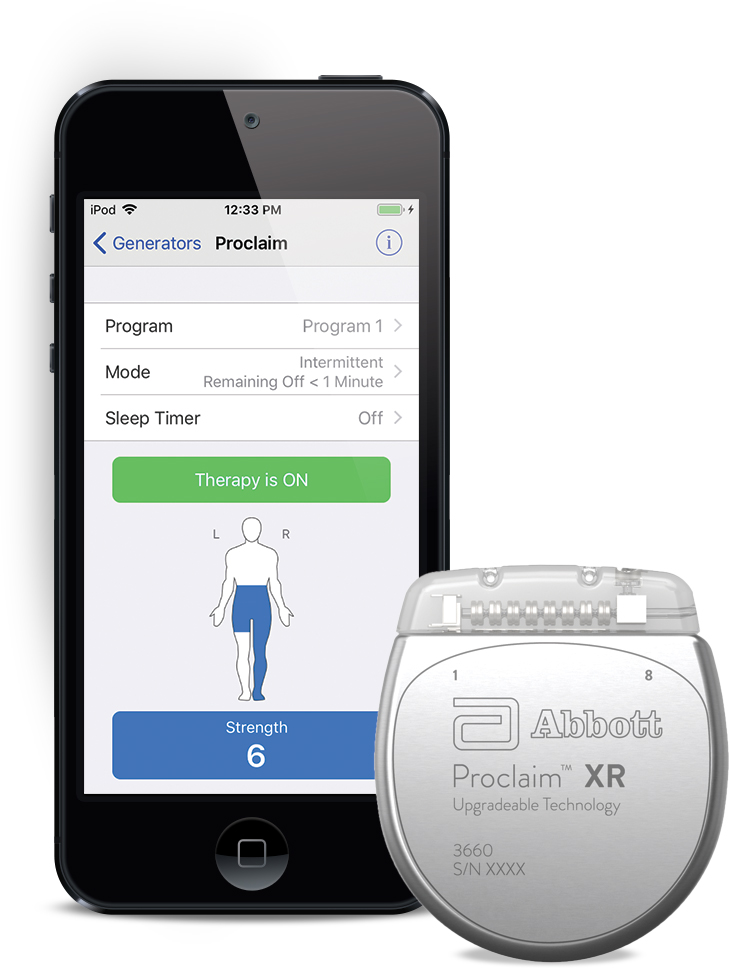
Abbott estimates that only about 0.06% of the recalled devices malfunctioned, with 0.03% of them resulting in loss of therapy and additional surgery. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
